Description
Product Information
Manufacturer: GE
Description
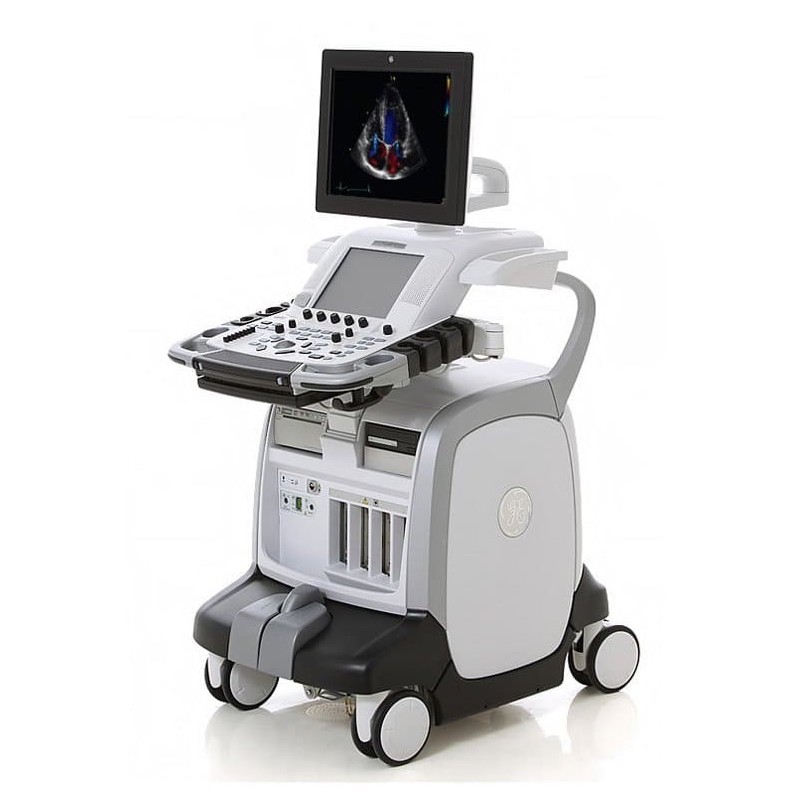
The GE Vivid E9 is the newest addition to GE Medicals long line of high quality ultrasound systems. GE’s exclusive, patented, beamforming technology; provides eight times the power of traditional ultrasound systems with increased volume size for full volume single beat 4D acquisition. Using both coherent and harmonic image processing, the system provides computational power, ease of imaging, workflow flexibility and product upgradeability.
Performance
- High resolution 17 inch High Definition LCD display
- 5 active probe ports
- Rear handle
- 2D & 4D tissue operating mode
- 2D & 4D color flow operating mode
- Color M-mode
- Tissue velocity M-mode
- Tissue M-mode
- Pulsed wave Doppler
- Anatomical M-mode
- Curved anatomical M-mode
- Tissue velocity imaging
- Tissue tracking
- Tissue synchronization imaging
- Strain imaging
- Strain rate imaging
- Tissue velocity Doppler
- Blood flow imagine
- Blood flow angio flow imaging
- Bi-plane, Tri-plane 4D option
- Bi- and- Tri-plane with color 4D option
- 4D stress
- 4D Strain Imaging optional
- CW Doppler
- Patient Information Database
- Image Archive on hard drive
- Comprehensive Cardiac Calculation package
- Comprehensive Vascular calculation package
- Stress echo
Capabilities
????
Available Image Probes
4C-D Wide Band Convex Probe, M5S-D Active Matric Single Crystal Phased Array Probe, 4V-D Active Matrix /4D Volume Phased Array Probe, 6S-D Phased Array Probe / Pediatric, 12S-D Phased Array Probe, 9L-D Linear Array Probe, 11L-D Linear Array Probe, ML6-15-D Linear Array Probe, I13l Curved Array Probe, P2D Pencil Probe, P6D Pencil Probe, 6T TEE Probe, 6Tc TEE Probe, 9T TEE Probe
or fill out our Contact Form.


Reviews
There are no reviews yet.